CBD Product Quality Assurance: Panaxia & Tikva
By Dr. Malgorzata Meunier
Product quality and patient safety are our priorities at Panaxia Pharmaceuticals. Our company fully complies with strict good manufacturing practices—GMPs, as they are often called. And as all pharmaceutical manufacturers should, we ensure GMPs through the continuous development and execution of quality assurance. This quality assurance and our following of GMPs mean that Tikva— a consumer CBD product brand supported by our manufacturing– are also supported by our procedures.
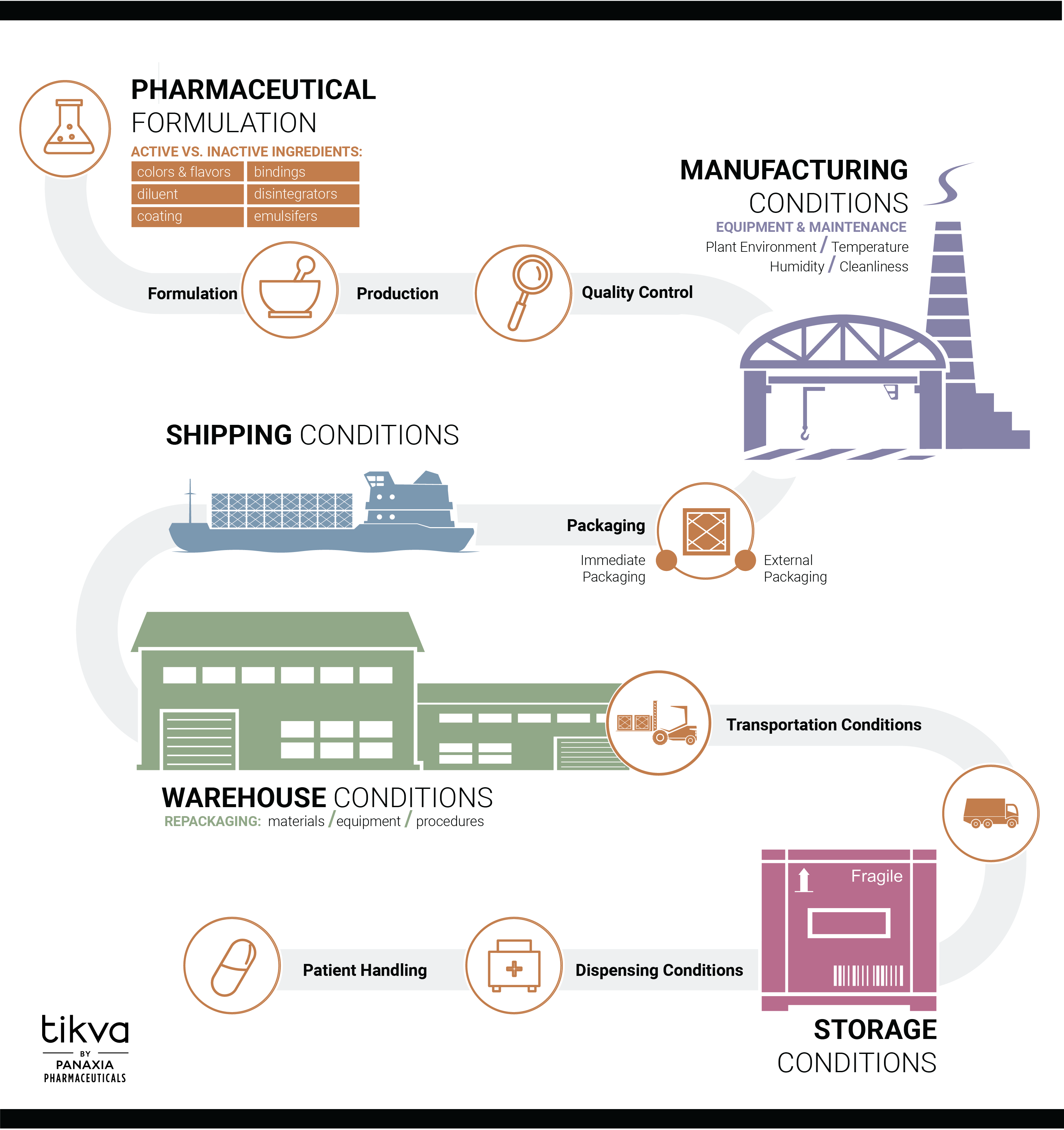
Panaxia knows that quality assurance is crucial for keeping consumers safe. Panaxia therefore works to ensure that all our manufactured CBD products delivered to a consumer are quality-controlled, safe, and effective. Panaxia applies quality assurance throughout all organizational areas. We run all activities according to our company’s quality policy.
Today, Panaxia’s quality assurance ensures that all its products meet GMP standards, including:
- Each product will consistently meet pre-determined quality specifications and requirements at all stages of development, manufacturing, storing, distribution, and sales.
- Applied materials are of the highest quality and come from qualified suppliers.
- High-level technology and sanitation are administered to all manufacturing rooms and equipment.
- Production is organized in a way to help reduce the risk of error during the manufacturing process.
- Qualified, regularly trained staff are employed throughout the manufacturing process.
- Quality control is conducted using the modern equipment and precise analytical methods at all stages of the manufacturing process.
- Quality control of all products and raw materials include testing and control through validated analytical methods around the following parameters:
- Microbial burden
- Toxin content
- Cannabinoids content and standardization of main cannabinoids
- Residual solvents
What this means for you, or others who are considering using Tikva hemp-derived CBD products, is that our products are supported by manufacturing processes that ensure consistency, accuracy, and an end product you can be confident in.
Dr. Malgorzata (Gosia) Meunier holds a Ph.D. in Pharmaceutical Sciences and was formerly acting as R&D Lead and Principal Scientist in several Swiss pharmaceutical companies, developing innovative drug formats. Dr. Meunier has been living in Israel for more than two years and has been working as Global Scientific Director in Panaxia Pharmaceuticals Ltd., in charge of the global business expansion of Panaxia’s innovative pharmaceuticals.
Panaxia Pharmaceuticals is an Israeli manufacturer of cannabinoids. The Company is part of a larger group of companies which have manufactured pharmaceuticals for more than 40 years. Together, the group produces more than 600 different conventional medication products in Israel, with a wide range of indications from sore throat lozenges to dermal fillers. The group sells into more than 40 countries, with a heavy focus on research and development. Outside the United States, Panaxia has been developing and manufacturing pharmaceutical products based on cannabis derivatives since 2010. To learn more, visit panaxia.co.il/
An additional article we think you will like -Tikva’s CBD and Melatonin.






