What makes Tikva’s product formulations special?
One of the most important aspects of an effective medicine is the mode of administration. Each mode, or delivery method, has specific purposes, advantages, and disadvantages. 
Routes of administration greatly affect bioavailability by changing the number of biologic barriers a drug must cross or by changing the exposure of a drug to pumping and metabolic mechanisms (Bardal et al., 2011). Bioavailability is the proportion of a drug or other substance that enters the blood circulation when introduced into the body and so is able to have an active effect. It is essential for a medical product to have high bioavailability to ensure the API (“active pharmaceutical ingredient,” in this case CBD) achieves the desired goal and reaches the desired part of the body. Modes of administration include oral (tablets, capsules, chewables, liquids), injection, sublingual, rectal, vaginal, ocular, otic, nasal, topical, and inhalation.
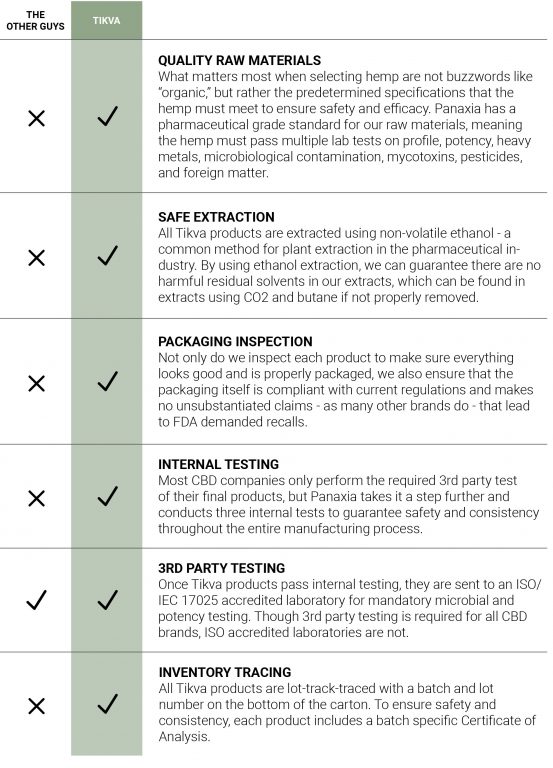
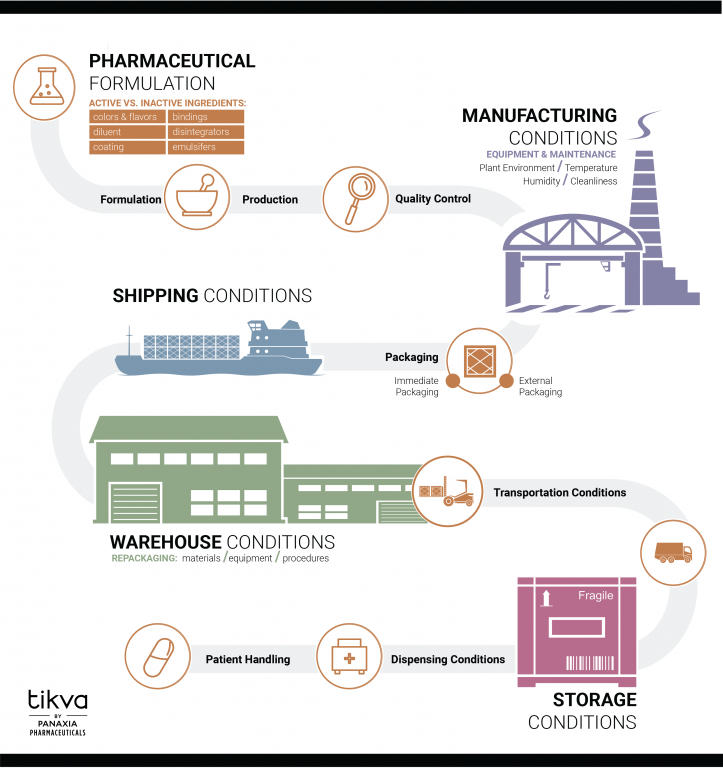
Unlike other cannabis companies, Panaxia supports Tikva’s products with pharmacokinetic data to know how fast and how completely the API is absorbed into the body. Pharmaceutical formulation development is a crucial step to determine the optimal dosage, composition, and process to achieve a manufacturable, chemically and physically stable throughout the manufacturing process and product shelf life, and bioavailable (i.e., it must contain the exact amount of API in each dose that can be readily absorbed by the human body) product. Also, many quality standards and special requirements must be met to ensure the product’s efficacy and safety.

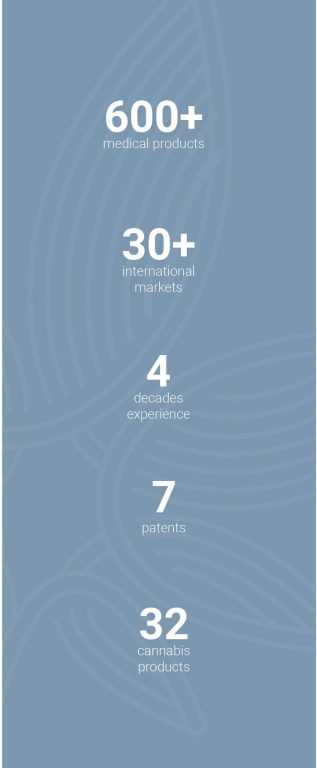
Panaxia currently holds seven patents on delivery methods and formulations.
Proprietary analytical methods allow Panaxia’s R&D team to specify and standardize up to nine components in Tikva products. They also can identify and quantify eleven major and minor cannabinoids, including THC, THCA, CBD, CBDA, CBN, CBG, CBGA, CBC, THCV, delta-8-THC and 11-hydroxy-THC. Tikva products are always made from full-spectrum hemp extracts, not CBD isolates or broad-spectrum extracts. Our formulations maximize absorption and bioavailability, which leads to consistently increased efficacy. The main raw plant materials and formulation excipients were selected to synergistically facilitate the functioning of the dominant plant actives (CBD).

Because Panaxia conducts these studies, they can ensure each Tikva delivery method effectively and efficiently delivers the CBD where and when needed. For example, Tikva has three types of tablets: CBD Oral Tablets, CBD + Melatonin Tablets, and CBD + Myo-inositol tablets. Our tablets are formulated to be absorbed directly into the bloodstream, so the API (CBD) avoids the “first pass effect” (metabolism by the liver). These tablets are not gel-caps, which are what are most commonly used in the CBD industry – unless gel-caps have been specifically formulated to increase bioavailability, these capsules will not be absorbed as effectively as Panaxia’s specially formulated powder tablets.

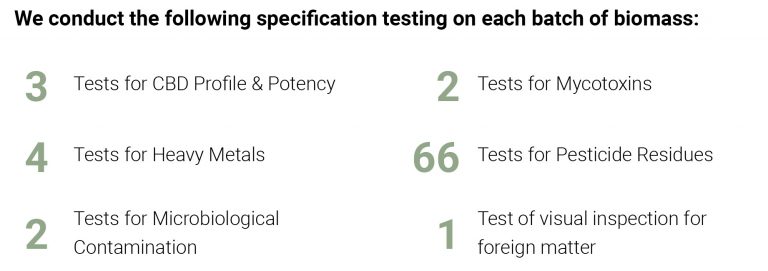
Along with Pharmacokinetic studies, Panaxia performs stability testing on all Tikva products to ensure safety and verify shelf-life duration. Stability testing provides evidence on how the quality of a drug substance or drug product varies with time under the influence of environmental factors such as temperature, humidity, and light, and to establish a re-test period and shelf-life. These tests are essential in traditional pharmaceuticals, and they should be applied to medical hemp and cannabis products as well.
You may have noticed that some other products have higher quantities of CBD per milligram compared to Tikva products. This is because Tikva products are designed to be absorbed effectively, so we don’t need to hyper-concentrate our formulations.

A high concentration cannabis oil product may say it has a high dose of CBD, but it could have very low bioavailability, making it less effective than a lower concentration oil with high bioavailability. From a pharmaceutical perspective, it is always recommended to start at the lowest dose and increase as needed. You should not be taking a higher dose than actually needed, which is often the case with these hyper-concentrated formulas. “Super-sized” high- dose oils are not necessarily more effective. A lower-dose product with higher bioavailability is more efficacious and better for you overall.
From a compliance standpoint, we have positioned our CBD products as dietary supplements in anticipation for when the FDA will designate CBD as such – while other brands will have to recall their products and re-design/re-print all their packaging to be compliant, Tikva has planned ahead in order to be one of the few brands that can remain on shelves when these new regulations commence, so you do not need to worry about inconsistent access or availability of Tikva products.